-
Amylyx Pharmaceuticals Announces Posting of Briefing Documents for Second FDA Meeting on AMX0035
-
Availability of Information Given to Members in with CDRH Open Panel Meetings; Draft Guidan
-

Fillable Online FDA Document Email Print -
-
![PDF] Ziprasidone for Injection 1 Advisory Briefing Document Appendix 1, The Behavioural Activity Rating Scale | Semantic PDF] Ziprasidone for Injection 1 Advisory Briefing Document Appendix 1, The Behavioural Activity Rating Scale | Semantic](https://d3i71xaburhd42.cloudfront.net/608dde6ea19d8f7b35c9876408fad41316408421/6-Table2-1.png)
PDF] Ziprasidone for Injection 1 Advisory Briefing Document Appendix 1, The Behavioural Activity Rating Scale | Semantic
-

Briefing Book Template - Fill Printable, Blank | pdfFiller
-
FDA Document - Pediatric Oncology of the Oncologic Drugs Advisory Committee (ODAC) June 17 and 18, 2020
-

Briefing Documents. Presentation What is a BD BD process Tips download
-
FDA Briefing Information for the August 13, 2020 Meeting of Oncologic Drugs Advisory Committee (AM
-
THE DEBRIEF ON BRIEFING HOW TO MAXIMIZE YOUR BRIEFING BOOK FOR FDA COMMITTEE MEETINGS
-
FDA Briefing Document - Spark Inc, LUXTURNATM: Cellular, Tissue, and Gene Therapies Advisory Committee Meeting - O
-
AVEED Undecanoate UCM348092 PDF | Trial |
-
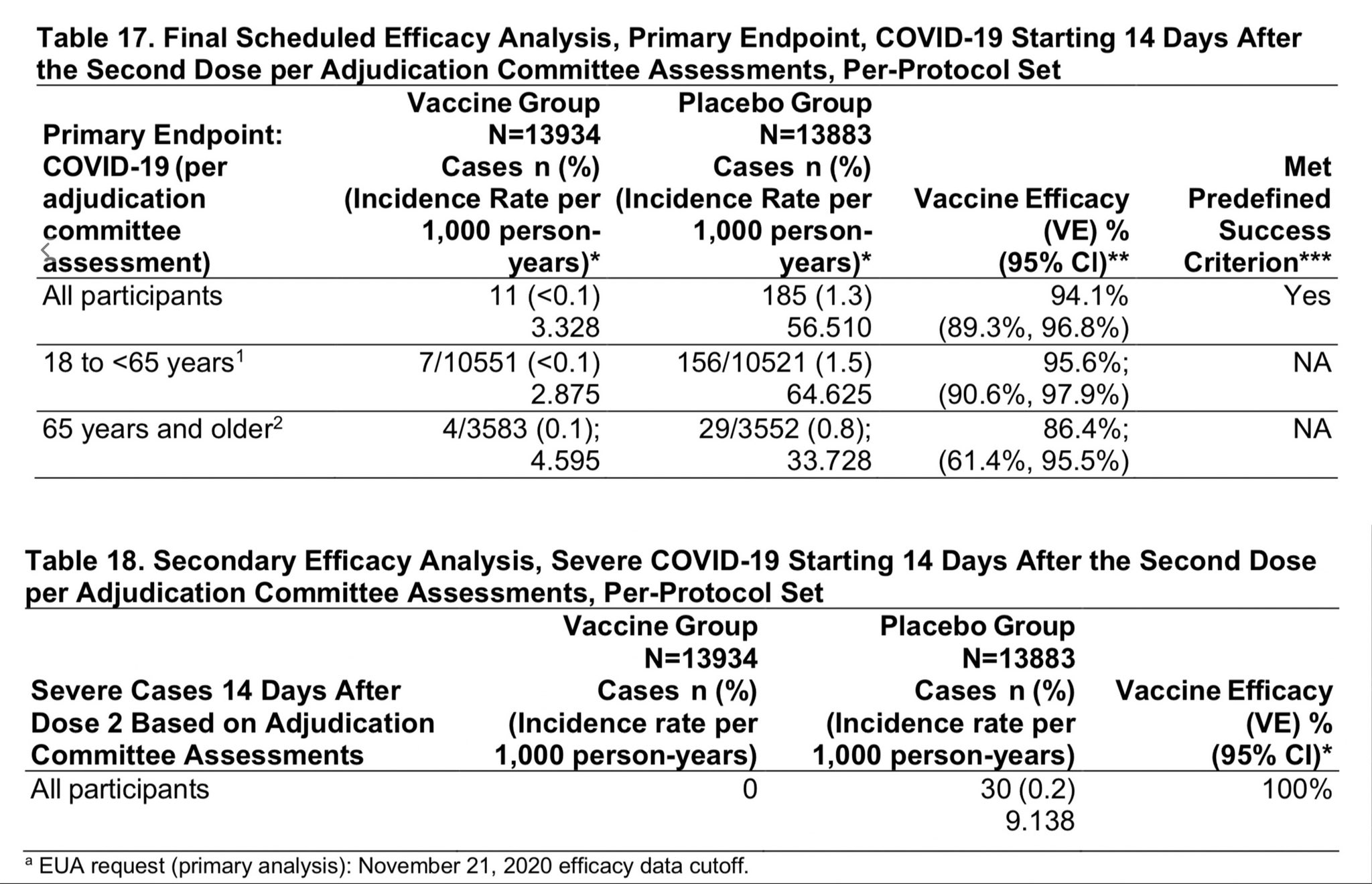
David R. Liu on Twitter: "Want to review #SARSCoV2 #COVID19 safety and efficacy data yourself? Linked below is the briefing document for the FDA vaccine committee on Moderna's mRNA
-

Briefing Documents. Presentation What is a BD BD process Tips download
-
November 9, 2022 Meeting of the Pulmonary-Allergy Drugs
-
Zealand announces FDA's posting of briefing documents for the Advisory and the lixisenatide/
-

Merck, ODAC Like US FDA's Point-Counterpoint Briefing Document :: Pink Sheet
-
FDA Briefing Document: Peripheral and Central Nervous System Drugs Committee Meeting
-
Amylyx Pharmaceuticals Posting of Briefing Documents for FDA Advisory Meeting on AMX0035
-
FDA ADVISORY COMMITTEE BRIEFING DOCUMENT MNK-812 JOINT MEETING THE AND ANALGESIC DRUG ADVISORY COMMITTE
-
-
Briefing Document Endocrinologic Drugs Advisory Committee Meeting
-
March 28-29, 2023 Joint Meeting of the Drug Safety and Risk Management Advisory Committee and Dermatologic and Drugs

